More information
2022/11/29
2022/11/29
2022/11/29
Application of silicone oil in lubricating oil
2019/03/11
Determination of isocyanate content
Published:
2019/08/13
The quantitative determination methods of isocyanate include gravimetric method (dry method) and volumetric method (wet method). The principle of both methods is based on the reaction of primary amine or secondary amine with isocyanate to generate corresponding urea, and the reaction is carried out quantitatively under the condition of excessive amine.
The quantitative determination methods of isocyanate include gravimetric method (dry method) and volumetric method (wet method). The principle of both methods is based on the reaction of primary amine or secondary amine with isocyanate to generate corresponding urea, and the reaction is carried out quantitatively under the condition of excessive amine. The difference between the two methods is that the gravimetric method is to separate the generated urea and calculate the content of isochlorite according to its weight (commonly known as dry method); Volumetric method is to use standard hydrochloric acid solution to drip back the excessive amine, and calculate the isocyanate content according to the consumption of hydrochloric acid. The former has complicated analysis steps, slow analysis speed and is rarely used. At present, the widely used isocyanate quantitative determination method mainly refers to volumetric method (commonly known as wet method). This method is based on acid-base reaction. If the sample contains free acid, this method is based on acid-base reaction. If the sample contains free acid, it must be corrected. The reaction principle is as follows:
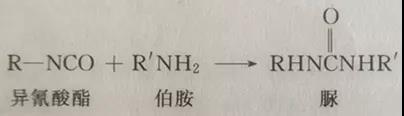
or
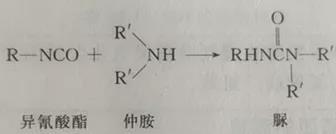
Excess amine reacts with hydrochloric acid to form organic amine hydrochloride:
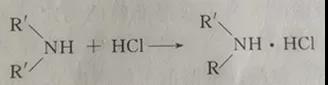
As a hydrogen donor reacting with isocyanate, the selection of amine has a greater impact on the analysis results. It is necessary to select fast reacting and nonvolatile amines, such as dibutylamine and piperidine (commonly known as hexahydropyridine), while aromatic amines and long-chain fatty amines react slowly. Isocyanate will react with water and alcohol. The solvent should be non-polar solvent such as toluene or chlorobenzene, trichlorobenzene, etc., and dehydration treatment should be carried out before use. The indicator is bromophenol blue or bromocresol green. The determination of isocyanate content is the most important production control index for both manufacturers and product factories. As isocyanate may be polluted by water and impurities during production, storage and transportation, resulting in a decrease in content, content analysis must be done to provide a basis for accurate formula calculation. The measurement steps are as follows.
1. Reagent preparation
(1) 2mol/L Di-n-butylamine (fumigated anhydrous), toluene (anhydrous) or chlorobenzene (anhydrous chlorobenzene dried by calcium chloride) solution.
(2) 0.5mol/L hydrochloric acid standard solution (preparation and calibration shall be conducted according to routine chemical analysis).
(3) 0.1% bromocresol green isopropanol (or methanol) solution; If bromophenol blue is selected as the indicator, first weigh 0.1g bromine
Phenol blue was co prepared with 1.5mL1mol/L NaOH aqueous solution and diluted to 100mL with water
2. Analytical procedures
Accurately weigh 2.5~3.5g isocyanate sample (liquid sample can be sampled with an ampere bulb) into a 250mL conical flask, add 10mL toluene (or chlorobenzene) for dissolution, measure 20mL dibutylamine solution with a pipette (calibrated), shake it to make it uniformly mixed, place it at room temperature for 15~30min, add 70~100mL anhydrous isopropanol (methanol or isopropanol) to dilute the sample, and then add 0.5mL bromocresol green (or bromocresol blue) indicator, Titrate with 0.5mol/L hydrochloric acid standard solution until the solution turns from blue to yellow for 15s and does not disappear. Carry out blank test with the same procedure.
3. Calculation results

Where, V blank - volume of hydrochloric acid standard solution consumed in blank test;
V Sample 1 Volume of hydrochloric acid standard solution consumed by the sample.
Generally, the content of hydrolyzed chlorine in refined isocyanate is 0.001% ~ 0.01%, and the analysis result of this method need not be corrected. The hydrolysis chlorine content of crude isocyanate is more than 0.05%, and the analysis results should be corrected.

Where 36.46 is the relative molecular weight of HC1.
4. Precautions
Isocyanate is easy to absorb water, so it is important to prevent moisture and active hydride from interfering in sampling; Toluene can be used as solvent for most refined isocyanates, and chlorobenzene or trichlorobenzene can be used for crude isocyanates.
keywords
:Next
:Next
Contact Us
Nantong San Ying Polyurethane Co., Ltd.
Address:8th floor, Hurunxingcheng, No. 555 Haiyang North Road, Chengbei Street, Rugao City, Jiangsu Province, China.
Tel:+86-513-87691698
Mobile:+86-18962773679
Fax:+86-513-88560029
Email: jane@sanychem.com
Products
Follow us


Copyright © 2022 Nantong San Ying Polyurethane Co., Ltd.